Bioburden Testing Market Share: Key Trends and Sector Insights
The landscape of microbiological safety is shifting as manufacturers prioritize high-speed accuracy in contamination control. Beyond simple compliance, the ability to command a significant Bioburden Testing Market Share now depends on the integration of rapid detection technologies and automated workflows. As the industry moves toward 2031, the competition for market dominance is intensifying among diagnostic giants and specialized laboratories.
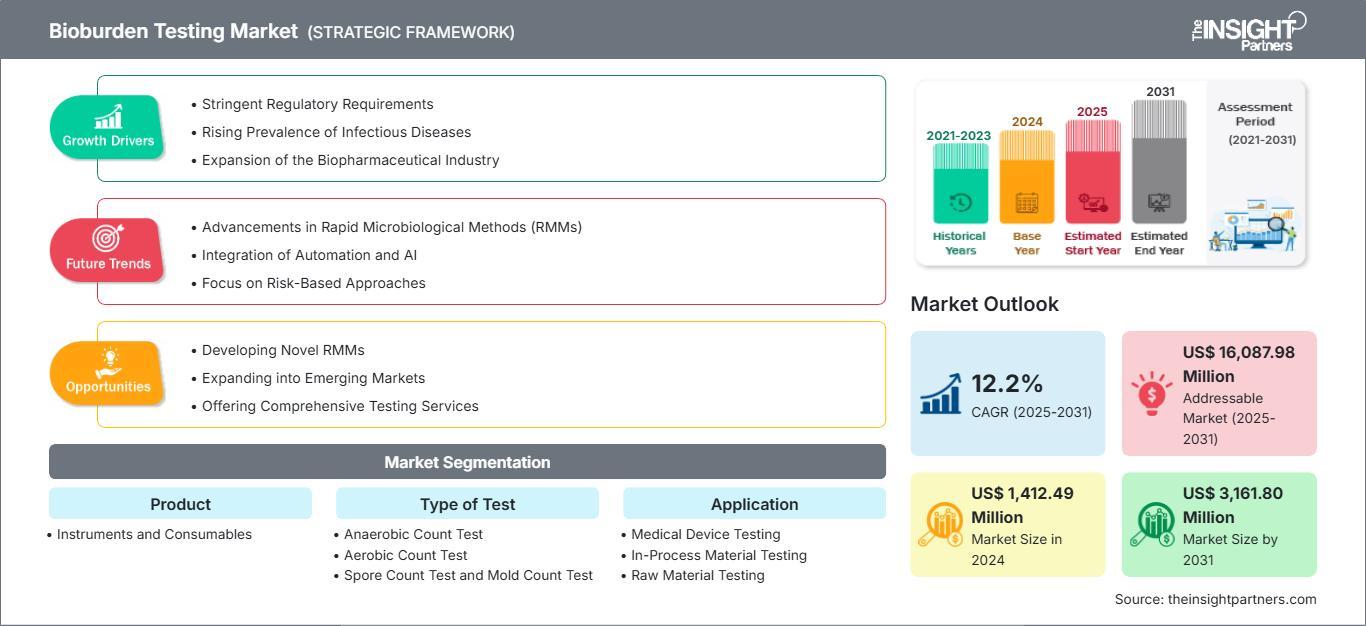
The Bioburden Testing Market size is expected to reach US$ 3,162 million by 2031. The market is anticipated to register a CAGR of 12.2% during 2025–2031. This growth is a reflection of the critical role that microbial enumeration plays in the safety of the modern healthcare supply chain.
Download Sample Report - https://www.theinsightpartners.com/sample/TIPRE00003000
Primary Market Report Drivers
The steady expansion of this sector is fueled by several structural drivers that influence how companies allocate their quality control budgets:
1. Dominance of the Pharmaceutical and Biotechnology Sector The pharmaceutical industry currently holds the largest portion of the Bioburden Testing Market Share. With the explosion of biologics and biosimilars, the risk of microbial contamination has become a multi-million dollar concern. Pharmaceutical giants are no longer just testing finished products; they are embedding bioburden assessments into every stage—from raw material screening to in-process monitoring—to ensure that high-value batches are not lost to unforeseen microbial growth.
2. Increasing Outsourcing to Contract Manufacturing Organizations (CMOs) A significant trend impacting the market is the shift toward outsourcing. Many medical device and drug manufacturers are moving away from in-house testing to lean on the expertise of specialized CMOs and third-party laboratories. This transition is driving the service-based Bioburden Testing Market Share, as these specialized facilities can offer higher throughput and more advanced validation protocols than many internal quality control departments.
3. Heightened Focus on Medical Device Sterility Assurance As surgical procedures become more complex and the use of minimally invasive tools rises, the "pre-sterilization" phase has gained new importance. Bioburden testing is the primary method used to determine the appropriate sterilization dose for medical equipment. This reliance ensures that as the medical device industry grows, the demand for aerobic and anaerobic count tests follows suit, solidifying the market's long-term revenue base.
Top Players Leading the Market
The competitive environment is highly concentrated, with a few key players holding a majority of the Bioburden Testing Market Share. these companies lead through constant innovation in consumables and the development of proprietary automated platforms:
- Thermo Fisher Scientific Inc.
- Merck KGaA
- bioMérieux SA
- BD (Becton, Dickinson and Company)
- QIAGEN
- SGS SA
- Pacific BioLabs
- Nelson Laboratories, LLC
- Charles River Laboratories
- STERIS
Future Outlook: Technology as a Differentiator
The "share of wallet" in this market is moving toward companies that can provide Rapid Microbiological Methods (RMMs). Traditional plate count methods, which require days of incubation, are being challenged by ATP bioluminescence and PCR-based systems that offer results in a fraction of the time.
Furthermore, the integration of artificial intelligence (AI) into microbial colony counting is set to redefine the Bioburden Testing Market Share in the coming years. Automated systems that use AI to distinguish between microbial species and background noise reduce human error and accelerate the release of critical medical products to the market.
Conclusion
The path to 2031 is defined by a 12.2% CAGR and a market valuation exceeding US$ 3.1 billion. As regulatory standards tighten and the complexity of therapeutic products increases, the Bioburden Testing Market Share will continue to be a vital metric for the healthcare industry's health and safety.
Related Report :
· Pharmaceutical Quality Control Market Overview, Growth, Trends, Research Report (2021-2031)
Contact Information -
Email: sales@theinsightpartners.com
Phone: +1-646-491-9876
Also Available in :
Korean German Japanese French Chinese Italian Spanish